This page provides specifications of knee specimen OKS004. This is a pilot specimen to go over the whole experimentation process.
Contents
- Specimen Characteristics
-
Experimentation
- Specimen
- Specimen Preparation
- Joint Imaging
- Joint Mechanics
-
Tissue Mechanics
- Specimen Preparation
-
Testing
- oks004-FMC-ACXX-01
- oks004-FMC-ACXX-02
- oks004-FMC-ACuX-01
- oks004-MCL-CXXX-01
- oks004-PAT-CXXX-01
- oks004-PAT-CuXX-01
- oks004-PAT-CXXX-02
- oks004-MNS-MPXX-01
- oks004-MNS-MPXX-02
- oks004-MNS-MCwX-01
- oks004-ACL-CXXX-01
- oks004-PCL-CXXX-01
- oks004-LCL-CXXX-01
- oks004-PTL-CXXX-01
- oks004-QUAT-CXXX-01
- oks004-TIB-MAXX-01
- oks004-TIB-MAuX-01
- oks004-TIB-MAXX-02
- oks004-TIB-LAXX-01
- oks004-TIB-LAuX-01
- oks004-TIB-LAXX-02
- Data Analysis
- Modeling & Simulation
Specimen Characteristics
Right knee
Gender: Female
Age: 46 years
Race: White
Height: 62 in. (1.575 m)
Weight: 120 lbs. (54.43 kg)
BMI: 21.9
Serologically tested: Yes
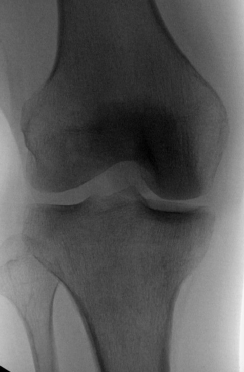
X-Ray:
Experimentation
-- aerdemir 2014-09-28 13:33:48 All, please complete Supporting Team Members, Timeline, and Data Location sections for the experimentation component you have led.
Specimen
Reference Specification: Specifications/Specimens - Revision as of April 28, 2014
Leading Team Member: Snehal Chokhandre
Supporting Team Members:
Timeline:
Data Location:
Protocol Deviations:
Specimen Characteristics. Donor age (46 yrs) does not match specifications.
Specimen Size. The specimen is not hip to toe rather mid-femoral shaft to toe.
Notes:
- None.
Specimen Preparation
Reference Specification: Specifications/SpecimenPreparation - Revision as of July 9, 2014
Leading Team Member: Tara Bonner
Supporting Team Members: Snehal Chokhandre, Craig Bennetts, Dylan Beckler, Katie Stemmer
Timeline:
- Specimen Thawing: 22.5 hours (12:30pm 7-20-14 to 11:00am 7-21-14)
- Specimen Dissection: 1.75 hours (12:45pm-2:30pm 7-21-14)
- Optotrak Base Plug Placement on Bones: 1.5 hours (2:35pm-4:10pm 7-21-14)
- Potting: 35 minutes (1:25pm-2pm 7-22-14)
Data Location:
- Patella Registration and Optotrak Marker Alignment:
- Sensor in state files are saved in main configuration folder for project
- The two data files are found in specimen configuration folder
- digitized_MRI_marker_state.cfg
- Patella_sensor_to_Reference.tdms
Protocol Deviations:
Femur Anatomical Landmarks. As the specimen did not include femoral head, landmarks on femoral shaft at its most proximal circumference were digitized.
Notes:
Registration Markers - Preparation. Drilling axis (for screw placement) and marker center are not concentric. Careful handling is necessary not to rotate the markers after their installation on bones. Preparation of the markers beforehand, by drilling on the lathe may be helpful to establish concentricity.
Registration Markers - Digitization. Digitization order for femur spherical markers are: 1 - anteromedial, 2 - medial, 3 - lateral. Digitization order for tibia spherical markers are: 1 - lateral, 2 - medial, 3 - posterior.
Specimen Dissection
- Mention that quad tendon must be isolating by removing fat around it and then kept moist with saline towel (suggest using tweezers and squeezing to remove muscle that is on tendon)
- Mention that four inch strip of tibia and fibula shafts must be completely cleaned 360 degrees around shaft and should be made as far proximal as possible without damaging joint capsule to allow room for plugs
- Add that fibula should be fixed to tibia for MRI
Optotrak Base Plug Placement on Bones
- From step 2: Specify that 3 plastic optotrak plugs are needed
- From step 5:
- Base plug for femur was placed laterally and for tibia was placed medially (deviation from specification).
- Oks004 had the 2 screw holes on posterior side instead of anterior side
- Mention if a screw shears off it is okay to have 2 full screws and a sheared one in the plug
- Mention if all screws shear then remove screws and try drilling bigger holes first (tip: test with non-cemented screws first to see if they shear)
- Suggest using k-wire in one hole to hold plug still when screwing in others
- Suggest checking that drill bit is short enough to prevent going through cartilage on patella (from section 3 of step 5)
- Mention cut vertical line in skin over patella and pull apart sides to make room for plug (from section 3 of step 5)
- From step 6: mention wiping off excess cement from screw before inserting
- General:
- have section on cement
- suggest making small batches and doing one plug at a time to avoid cement hardening before you are ready
- mention that if cement clumps up when screwing in then it is time for new batch
- Add caution that cement should be made in a hood and masks should be worn
- have section on cement
Registration Marker Placement on Femur and Tibia
- From step 1: mention that it is being driven by field of view from MRI spec
- General: Specify drill bit size, specify type of retractor (mention to check that it is sharp for grip), mention that tissue must be cut to make room for spheres
Preparation of Specimen for Mechanical Joint Testing
- Oks004 fibula was screwed/fixed to tibia at this point
- suggest checking that sensors will fit on plugs with the pot before actually potting
- mention removing wood metal that hardens over edge of pot with chisel
- mention liquid nitrogen is used to secure tendon
- mention that chuck should be used to protect knee when pouring liquid nitrogen onto tendon
- 0ks004 liquid nitrogen was added to tendon every 15-20 minutes
- consider smaller grommet size
- explain that size of sensor is determined by if it is small enough to fit in knee and large enough to collect all data
- From step 4 mention to check during dissection if any important features were cut when incisions were made
Joint Imaging
Specimen Preparation
Reference Specification: Specifications/SpecimenPreparation - Revision as of July 9, 2014
Leading Team Member: Craig Bennetts
Supporting Team Members: Snehal Chokhandre
Timeline:
Data Location:
Protocol Deviations:
- Patella registration marker assembly was not placed on the patella for imaging. This can be prevented by cross-checking with specifications. Alternative imaging or digitization is necessary to align patella local coordinate systems between imaging and motion analysis. This can be accomplished by i) imaging patella only with the registration marker assembly after dissection, or ii) by digitizing patella anatomy using Optotrak along with patella motion analysis markers. Snehal will follow up to resolve this issue.
- As an alternative solution to obtain the transformation matrix between the patella Optotrak marker coordinate system to image coordinate system, 48 points on the anterior bony surface of the patella was digitized using the Optotrak probe. First (and last) three points correspond to lateral, medial, and inferior points, respectively.
Notes: -- colbrunn 2016-10-13 16:50:01 In reviewing the imaging it appears the specimen moved slightly for the general purpose imaging set. No difference in anatomy location was identified between cartilage and other imaging sets. However, the general purpose imaging set was slightly different than the other 4 sets. Caution should be used when segmenting with the general purpose image set.
Imaging
Reference Specification: Specifications/ExperimentationAnatomicalImaging - Revision as of June 25, 2014
Leading Team Member: Craig Bennetts
Supporting Team Members: Snehal Chokhandre, Katie Stemmer
Timeline:
Data Location:
Data was received from the imaging facilities and uploaded to in-house data management server (http://cobicore.lerner.ccf.org/midas; only accessible within Cleveland Clinic network). Raw DICOM files were stored in the folder Open Knee(s) --> Private --> oks004 --> MRI --> DICOM
DICOM files were converted to NIfTI files, stored in the folder Open Knee(s) --> Private --> oks004 --> MRI --> NIFTI of the in-house data management server. NIfTI files were also disseminated to the public in the 'Downloads' section of the project website, https://simtk.org/home/openknee, under the package oks004.
Data storage and dissemination were accomplished based on the updated imaging specifications, see Revision as of April 16, 2015.
DICOM files were also split into series and uploaded to in-house data management server (http://cobicore.lerner.ccf.org/midas; only accessible within Cleveland Clinic network); in the folder Open Knee(s) --> Private --> oks004 --> MRI --> DICOM_SPLIT
- SEQUENCE 2: General Purpose Imaging
- t1_f13d_sag_p2_150_0.4_we
- Position R1.5 A20.3 F4.4
- t1_f13d_sag_p2_150_0.4_we
- SEQUENCE 3: Cartilage Imaging
- t1_f13d_sag_350x350x700_fs
- Position R1.5 A20.3 F4.4
- t1_f13d_sag_350x350x700_fs
- SEQUENCE 4: Connective Tissue Imaging
- pd_tse_sag_1.4mmslice_1avg_axial
- Position L0.7 A21.8 F1.5
- pd_tse_sag_1.4mmslice_1avg_sag
- Position L2.9 A18.2 H8
- pd_tse_sag_1.4mmslice_1avg_coronal
- Position L.2.9 A18.2 H8
- pd_tse_sag_1.4mmslice_1avg_axial
Protocol Deviations:
- After localizer we ran ACRT1 Axial test (a localizer/scout)
Notes:
- General purpose imaging seems very noisy; data source SEQ1_t1_fl3d_sag_iso_0.4_we (@ staging server for data organization)
- Cartilage imaging seems acceptable; data source SEQ2_t1_fl3d_sag_350x350x700_fs_11 (@ staging server for data organization)
- Connective tissue imaging (axial) seems acceptable; data source SEQ3_pd_tse_sag_1.4mmslice_1avg_axial (@ staging server for data organization)
- Connective tissue imaging (sagittal) seems acceptable; data source SEQ4_pd_tse_sag_1.4mmslice_1avg_sagittal_10 (@ staging server for data organization)
- Connective tissue imaging (coronal) seems to be a repetition of sagittal scan; data source SEQ5_pd_tse_sag_1.4mmslice_1avg_coronal (@ staging server for data organization)
- Carl Winalski, MD briefly evaluated the images provided herein; his important comments are provided below:
- The reconstructions on the 3D FLASH sequence shows a banding artifact. This is likely because of the way the images are scaled during the initial image production.
- The FOV looks rather large, raising concern about in-plane resolution, but maybe this is what is needed and the acquisition was adjusted by using a large matrix.
- The specimen has extensive bone infarctions. This is very unusual, but can be seen following high dose steroid treatment such as for bone marrow transplantation. This may not be good for use as a “normal” or simple “degeneration.”
- The high signal in the quadriceps and patellar tendons is from magic angle effect (tendon oriented 55 degrees to main magnetic field). Different positioning may accommodate this issue.
Joint Mechanics
Equipment Preparation
Reference Specification: Specifications/PressureCalibration - Revision as of July 17, 2014
Leading Team Member: Jason Halloran
Supporting Team Members: Robb Colbrunn, Katie Stemmer
Data Location:
Timeline:
Protocol Deviations:
- None.
Notes:
- Mention that equilibration might have to be run twice in order to successfully equilibrate sensor
Specimen Preparation
Reference Specification: Specifications/SpecimenPreparation - Revision as of July 9, 2014
Leading Team Member: Tara Bonner
Supporting Team Members: Snehal Chokhandre, Craig Bennetts, Dylan Beckler, Ahmet Erdemir
Timeline:
Data Location:
Protocol Deviations:
- None.
Notes:
- The specifications did not explicitly detail securing of the fibula to the tibia. A drill bit was passed through the fibula and the tibia to prevent fibula's movements during tibiofemoral joint testing. It may be necessary to secure fibula to the tibia during specimen preparation (before detaching the foot) to approximate anatomical alignment. This will require MRI compatible screws, etc. and will ensure keeping the fibula at the same position relative to the tibia in imaging and during robotics testing.
Testing
Reference Specification: Specifications/ExperimentationJointMechanics - Revision as of May 25, 2014
Leading Team Member: Tara Bonner
Supporting Team Members: Snehal Chokhandre, Craig Bennetts, Katie Stemmer, Jason Halloran, Ahmet Erdemir
Data Location:
Data was received from the robotics testing facility and uploaded to in-house data management server (http://cobicore.lerner.ccf.org/midas; only accessible within Cleveland Clinic network). Zipped folders were stored in the folder Open Knee(s) --> Private --> oks004 --> JointMechanics under relevant joint related folders.
Zipped folders were extracted and organized in the dissemination folder under Open Knee(s) --> Private --> oks004 --> JointMechanics --> DISSEMINATION of the in-house data management server. DISSEMINATION folder was downloaded and disseminated to the public in the 'Downloads' section of the project website, https://simtk.org/home/openknee, under the package oks004.
Data storage and dissemination were accomplished based on the updated joint mechanics testing specifications, see Revision as of October 29, 2015.
Timeline:
Protocol Deviations:
- For tibiofemoral joint testing, the knee was assembled upside down. This approach circumvented the need for gravity compensation during force measurements. The tibia was fixed directly to the load transducer, which was assembled on the testing frame.
- For patellofemoral joint testing, 20N compressive force for tibiofemoral joint was applied to increase stability
Notes:
- For tibiofemoral joint testing, reprogramming of the robotics testing system was required to accommodate optimization of the joint coordinate system.
- During tibiofemoral joint testing, the control system had issues loading the knee joint flexed at 90 degrees with a 2.5 Nm valgus moment from an unloaded state. This may be related to a possible singularity of the robot configuration.
- During tibiofemoral joint testing at 90 degrees flexion, visibility of the femur Optotrak markers was problematic.
- During patellofemoral joint testing, the knee needed to be oriented axially to accommodate loading direction of the quadriceps tendon. This caused the loss of tibia Optotrak marker data. Robotic system provides redundant information to reconstruct relative locations of patella, femur, and tibia.
- For patellofemoral joint testing, measurement of quadriceps line of action needs to be detailed.
- For patellofemoral joint pressure measurements, pressure data was collected to denote medial/lateral, superior/inferior locations of the sensor.
- During patellofemoral joint testing, patellofemoral kinematics data were not recorded for 0, 15, and 30 degrees flexion conditions. These conditions were repeated. In both case cases (initial and repeat) pressure data were collected.
- Knee should be aligned obliquely to accommodate line of action of quad because tibia markers are lost and data is not seen in camera
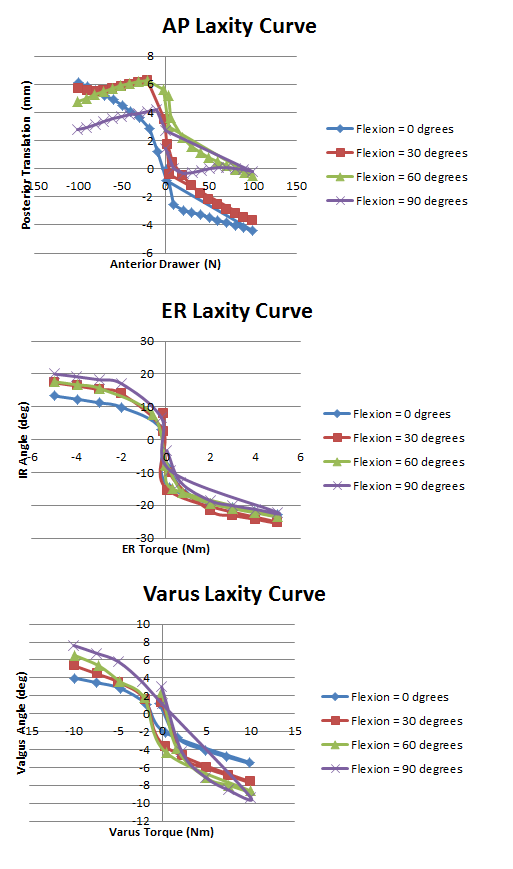
- During laxity testing at higher flexion angles (30, 60 and 90) non-zero external rotation torque caused the "Anterior Drawer" laxity condition to not truly be an anterior drawer test. The AP translations were not proportional to the AP loads due to coupling with ER rotation. In the future, attention should be made to ensure off-axis loads are zero during laxity testing. Gains and/or settling time can be increased to ensure this.
- Version of simVITRO software used was unreleased revision 3024 of the repository. The files can be found in svn://dev.lerner.ccf.org/data1/svn/repos/colbrunn-svn/Projects/Numbered Projects/2013CB-031 Open Knee/Original simVITRO-Tested on oks001 oks002 oks003 oks004
Tissue Mechanics
Specimen Preparation
Reference Specification: Specifications/SpecimenPreparation - Revision as of July 9, 2014
Leading Team Member: Snehal Chokhandre
Supporting Team Members: Craig Bennetts
Timeline:
Data Location:
Protocol Deviations:
- None.
Notes:
- None.
Testing
Reference Specification:
Leading Team Member: Snehal Chokhandre
Supporting Team Members:
Data Location:
Timeline:
Protocol Deviations:
- None.
Notes:
- The knee joint was dissected by a fellow. Observations during dissection noted no apparent injuries or damage to knee structures and cartilage.
- Same compression sample is used for both unconfined and confined compression. Sample lable ends with -01. Data label ends with 01 for unconfined and 02 for confined compression.
-- aerdemir 2014-09-28 13:31:28 Snehal, there seems to be a confusion in here in regard to your note about unconfined and confined compression. Are oks004-FMC-ACXX-01 and oks004-FMC-ACXX-02 different samples? If not shouldn't the sample name be oks004-FMC-ACXX-01 and unconfined compression data label be oks004-FMC-ACXX-01-01 an confinded compression data label be oks004-FMC-ACXX-01-02; see below. The headings below should reflect sample labels NOT data labels.
-- aerdemir 2014-09-28 13:42:44 Snehal, some of the labeling of samples do not conform our specifications, see Specifications/DataManagement. In particular tibia cartilage is referred as TIB (should be TBC), patella cartilage is referred as PAT (should be PTC), and quadriceps tendon is referred as QUAT. Please relabel all the samples in the freezer and your collected data to make sure we follow specifications.
oks004-FMC-ACXX-01
Trochlear groove, unconfined compression |
Diameter: 5 mm |
|
|
Thickness: 2.457 mm |
|
|
DAQ : 100Hz |
|
|
No video |
|
|
Room tempterature |
|
oks004-FMC-ACXX-02
Trochlear groove, confined compression |
Diameter: 5 mm |
|
|
Thickness: 2.457 mm |
|
|
DAQ : 100Hz |
|
|
No video |
|
|
Room tempterature |
|
oks004-FMC-ACuX-01
Trochlear groove, tensile test |
Length: 5.7 mm |
|
|
Width: 1 mm |
|
|
Thickness:0.8 mm |
|
|
DAQ: 100Hz |
|
|
Video: 5 Hz (Downsampled from 10 Hz) |
|
|
Room temperature |
|
|
3, 6, 9 % used (Deviation, user error) |
|
oks004-MCL-CXXX-01
MCL, tensile |
Length: 9.7 mm |
|
|
Width: 2 mm |
|
|
Thickness: 0.89 mm |
|
|
DAQ: 100 Hz |
|
|
Video: 2 Hz (user error) |
|
oks004-PAT-CXXX-01
Patella, unconfined compression |
Diameter: 5 mm |
|
|
Thickness: 2.06 mm |
|
|
DAQ: 100 Hz |
|
|
No Video |
|
oks004-PAT-CuXX-01
Patella, tensile test |
Length: 5.5 mm |
|
|
Width: 1 mm |
|
|
Thickness: 0.81 mm |
|
|
DAQ: 100 Hz |
|
|
Video: 5 Hz (Downsampled from 10 Hz) |
|
oks004-PAT-CXXX-02
Patella, unconfined compression |
Diameter: 5 mm |
|
|
Thickness: 2.06 mm |
|
|
DAQ: 100 Hz |
|
|
No Video |
|
oks004-MNS-MPXX-01
medial meniscus, unconfined compression |
Diameter: 5mm |
|
|
Thickness: 3.364 mm |
|
|
DAQ: 100 Hz |
|
|
No Video |
|
oks004-MNS-MPXX-02
medial meniscus, confined compression |
Diameter: 5mm |
|
|
Thickness: 3.364 mm |
|
|
DAQ: 100 Hz |
|
|
No Video |
|
oks004-MNS-MCwX-01
medial meniscus, tensile test |
Length: 6.4 mm |
|
|
Width: 1 mm |
|
|
Thickness: 0.716 mm |
|
|
DAQ: 100 Hz |
|
|
Video: 5 Hz (Downsampled from 10 Hz) |
|
oks004-ACL-CXXX-01
ACL, tensile test |
Length: 12.47 mm |
|
|
Width: 2 mm |
|
|
Thickness: 1.34 mm |
|
|
DAQ: 100 Hz |
|
|
Video: 5 Hz (Downsampled from 10 Hz) |
|
|
(Note: did not zero load) |
|
oks004-PCL-CXXX-01
PCL, tensile test |
Length: 12.257 mm |
|
|
Width: 2 mm |
|
|
Thickness: 1.23 mm |
|
|
DAQ: 100 Hz |
|
|
Video: 5 Hz (Downsampled from 10 Hz) |
|
|
(Note: sample was kept out overnight after |
|
|
testing,removed from system next morning) |
|
oks004-LCL-CXXX-01
LCL, tensile test |
Length: 11.5 mm |
|
|
Width: 2 mm |
|
|
Thickness: 1.23 mm |
|
|
DAQ: 100 Hz |
|
|
Video: 5 Hz |
|
oks004-PTL-CXXX-01
Patellar lig, tensile test |
Length: 14.5 mm |
|
|
Width: 2 mm |
|
|
Thickness: 1.138 Hz |
|
|
Video: 5 Hz |
|
oks004-QUAT-CXXX-01
Quad tendon, tensile test |
Length: 13.61 mm |
|
|
Width: 2 mm |
|
|
Thickness: 1.12 mm |
|
|
DAQ: 100 Hz |
|
|
Video: 5 Hz |
|
|
Note: sw crashed, test redone |
|
oks004-TIB-MAXX-01
Tibia cartilage, medial, unconfined compression |
Diameter: 5mm |
|
|
Thickness: 1.88 mm |
|
|
DAQ: 100 Hz |
|
|
No Video |
|
oks004-TIB-MAuX-01
Tibia cartilage, tensile, medial |
Length: 5.2 mm |
|
|
Width: 1 mm |
|
|
Thickness: 0.249 mm |
|
|
DAQ: 100 Hz |
|
|
Video: 5 Hz |
|
|
sw crashed, sample broke, new sample was taken |
|
|
and tested next day, strip was kept in the fridge |
|
oks004-TIB-MAXX-02
Tibia cartilage, medial, confined compression |
Diameter: 5mm |
|
|
Thickness: 1.88 mm |
|
|
DAQ: 100 Hz |
|
|
No Video |
|
oks004-TIB-LAXX-01
Tibia cartilage, lateral, unconfined compression |
Diameter: 5mm |
|
|
Thickness: 1.9 mm |
|
|
DAQ: 100 Hz |
|
|
No Video |
|
oks004-TIB-LAuX-01
Tibia cartilage, tensile, lateral |
Length: 6.67 mm |
|
|
Width: 1 mm |
|
|
Thickness: 0.82 mm |
|
|
DAQ: 100 Hz |
|
|
Video: 5 Hz |
|
|
Note: sw crashed, test repeated, may need to be |
|
|
repeated |
|
oks004-TIB-LAXX-02
Tibia cartilage, lateral, confined compression |
Diameter: 5mm |
|
|
Thickness: 1.9 mm |
|
|
DAQ: 100 Hz |
|
|
No Video |
|
Data Analysis
Registration
Note that the patella registration was not completed for this specimen. Registration code was modified to eliminate the patella registration information that was absent from the data set.
Spherical Markers
- Femur
<Anterior-Medial>oks004_MRG_FMR-M_AGS_01.stl
<Lateral>oks004_MRG_FMR-L_AGS_01.stl
<Medial>oks004_MRG_FMR-P_AGS_01.stl
- Tibia
<Posterior>oks004_MRG_TBR-P_AGS_01.stl
<Lateral>oks004_MRG_TBR-L_AGS_01.stl
<Medial>oks004_MRG_TBR-M_AGS_01.stl
- Patella
<Superior>oks004_MRG_PTR-S_AGS_01.stl
<Lateral>oks004_MRG_PTR-L_AGS_01.stl
<Medial>oks004_MRG_PTR-M_AGS_01.stl
Digitizing Order
- Tibia:
- Sphere 1 = Lateral
- Sphere 2 = Medial
- Sphere 3 = Posterior
- Femur:
- Sphere 1 = Anterior-Medial
- Sphere 2 = Medial
- Sphere 3 = Lateral
Joint Mechanical Testing
Pictured below are the Anterior Posterior laxity graph, IR/ER laxity graph, and the Varus/Valgus laxity graph.
Note: After further examination, it became clear that the posterior drawer test was not a pure laxity test.