This page provides specifications of knee specimen OKS006. This is a (likely healthy) elderly female specimen candidate.
Specimen Characteristics
Right knee
Gender: Female
Age: 71 years
Race: White
Height: 1.524 m
Weight: 49.4 kg
BMI: 21.3
Serologically tested: Yes.
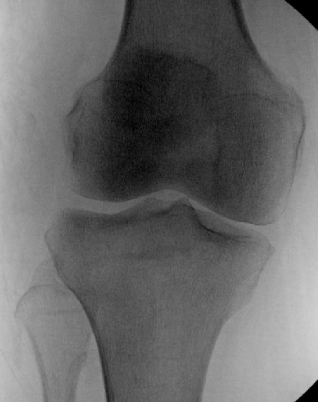
X-ray:
Experimentation
-- aerdemir 2015-01-02 17:16:12 All, please complete relevant sections of this page during and after experimentation.
Specimen
Reference Specification: Specifications/Specimens - Revision as of January 4, 2015
Leading Team Member: Snehal Chokhandre
Supporting Team Members: Ahmet Erdemir
Timeline: January 4, 2015 @ ~ 9:30 AM (specimen pulled out of freezer)
Data Location:
Protocol Deviations:
- None.
Notes:
- None.
Specimen Preparation
Reference Specification: Specifications/SpecimenPreparation - Revision as of January 5, 2015
Leading Team Member: Tara Bonner
Supporting Team Members: Snehal Chokhandre, Craig Bennetts, Ahmet Erdemir
Timeline:
- Thawing: ~9:30 AM January 4, 2015 - ~10:30 AM January 5, 2015
- Dissection: ~10:45 AM - ~11:30 AM January 5, 2015
- Assembly of Optotrak Base Plugs: ~ 12:30 PM - ~1:30 PM January 5, 2015
- Assembly of Registration Markers: ~1:30 PM - ~2.15 PM January 5, 2015
- Probing of Anatomical Landmarks: ~ 2:30 PM - ~3:30 PM January 5, 2015
- Probing of Registration Markers: ~ 2:30 PM - ~3:30 PM January 5, 2015
Data Location:
Protocol Deviations:
- Femur registration markers were assembled medially, laterally, and posteriorly (rather than anteriorly or anteromedially). Registration markers were digitized in the following order: medial, lateral, posterior. This was done to accommodate the quadriceps tendon.
Notes:
- Specimen did not thaw (at the thigh) adequately. The team waited an additional hour for dissection.
- Dissected and keeping skin-fat-muscle layers from thigh and shank.
- Dissected and keeping the femoral head.
- Dissected and keeping bone from femoral and tibial shafts.
Joint Imaging
Specimen Preparation
Reference Specification: Specifications/SpecimenPreparation - Revision as of January 5, 2015
Leading Team Member: Craig Bennetts
Supporting Team Members: Snehal Chokhandre
Timeline: ~3:30 PM - 4:00 PM January 5, 2015 (patella registration marker will be placed next day)
Data Location:
Protocol Deviations:
- None.
Notes:
- None.
Imaging
Reference Specification: Specifications/ExperimentationAnatomicalImaging - Revision as of January 6, 2015
Leading Team Member: Craig Bennetts
Supporting Team Members: Snehal Chokhandre, Ahmet Erdemir
Timeline:
- Specimen Placement in Transportation Fixture: ~9:30 AM - ~10:30 AM January 6, 2015
- Imaging: ~11:00 AM - ~1:00 PM January 6, 2015
Data Location:
Data was received from the imaging facilities and uploaded to in-house data management server (http://cobicore.lerner.ccf.org/midas; only accessible within Cleveland Clinic network). Raw DICOM files were stored in the folder Open Knee(s) --> Private --> oks006 --> MRI --> DICOM
DICOM files were converted to NIfTI files, stored in the folder Open Knee(s) --> Private --> oks006 --> MRI --> NIFTI of the in-house data management server. NIfTI files were also disseminated to the public in the 'Downloads' section of the project website, https://simtk.org/home/openknee, under the package oks006.
Data storage and dissemination were accomplished based on the updated imaging specifications, see Revision as of April 22, 2015.
Protocol Deviations:
- None.
Notes:
- General purpose imaging
- Cartilage imaging
- Connective-tissue imaging
Joint Mechanics
Equipment Preparation
Reference Specification: Specifications/PressureCalibration - Revision as of January 6, 2015
Leading Team Member: Robb Colbrunn
Supporting Team Members: Ahmet Erdemir
Timeline: ~10:00 AM - ~11:00 AM January 6, 2015
Data Location:
Protocol Deviations:
- None.
Notes:
- Sensor model is 5051, label is 5051-P1-16813DT1-1200.
- A single pass equilibration at 6 bars was conducted.
- A double pass equilibration was conducted at an additional pressure level of 3 bars, by using single pass 6 bars data. Software reordered the passes.
- Both equilibrations were repeated due to a sensel equilibration issue at low pressure (3 bar). A snapshot of the equilibration measurements were taken at 3 bar. Files from this repetition (in particular for double pass) were used for calibration.
- Versions of software used during testing
- simVITRO 1.0.3.12
- simVITRO Knee Module 1.0.4.8
- SSCAD Toolkit 1.0.3.18
- SSCAD Toolkit Extensions 1.0.1.3
- SSCAD NDI Optotrak 1.0.0.8
- NDI Optotrak Drivers 1.0.0.6
BioRobotics Software Configuration 1.0.1.5
BioRobotics Common Tools 1.0.6.3
Specimen Preparation
Reference Specification: Specifications/SpecimenPreparation - Revision as of January 6, 2015
Leading Team Member: Tara Bonner
Supporting Team Members: Robb Colbrunn
Timeline:
- Specimen preparation for TFJ testing: ~1:00 PM - ~2:00 PM January 6, 2015
- Specimen preparation for PFJ testing: ~7:30 AM - ~9:00 AM January 7, 2015
Data Location:
Protocol Deviations:
- Fibula was fixed to tibia with a wood screw immediately after imaging before potting. This practice was adapted for oks003 and other previous specimens in various forms. The specifications may need to change to reflect this.
Notes:
- None.
Testing
Reference Specification: Specifications/ExperimentationJointMechanics - Revision as of January 6, 2015
Leading Team Member: Tara Bonner
Supporting Team Members: Dylan Beckler
Timeline:
- Specimen Mounting for TFJ testing: ~2:00 PM January 6, 2015
- Testing of TFJ: ~3:00 PM - ~5:45 PM January 6, 2015
- Specimen Mounting for PFJ testing: ~9:00 AM January 7, 2015
- Testing of PFJ: ~ 12:00 PM - ~1:00 PM January 7, 2015
Data Location:
Data was received from the robotics testing facility and uploaded to in-house data management server (http://cobicore.lerner.ccf.org/midas; only accessible within Cleveland Clinic network). Zipped folders were stored in the folder Open Knee(s) --> Private --> oks006 --> JointMechanics under relevant joint related folders.
Zipped folders were extracted and organized in the dissemination folder under Open Knee(s) --> Private --> oks006 --> JointMechanics --> DISSEMINATION of the in-house data management server. DISSEMINATION folder was downloaded and disseminated to the public in the 'Downloads' section of the project website, https://simtk.org/home/openknee, under the package oks006.
Data storage and dissemination were accomplished based on the updated joint mechanics testing specifications, see Revision as of August 25, 2015.
Protocol Deviations:
- None.
Notes:
In tibiofemoral joint testing, during combined loading at 30 degrees flexion, the testing ended prematurely with abrupt loading. The test was repeated, in following the scheduled reproducibility testing was conducted and visually compared to first reproducibility testing. Any differences (<0.5 mm translation at peak loads) were attributed to specimen conditioning (and possibly creep).
In tibiofemoral joint testing, for combined loading at 90 degrees flexion, the knee fixture (holder angle) was rotated to accommodate range of motion of the robot. SimVitro takes into account of these adjustments to provide desired loading scenarios.
- In tibiofemoral joint testing, an optimized joint coordinate system was calculated from passive flexion data (0 to 90 degrees) under 50 N compression load (not 100 N). This was the case for all specimens so far and the specifications should reflect that.
- In patellofemoral joint testing, optimized tibiofemoral joint coordinate system was always re-established from passive flexion data (0 to 60 degrees) under 50 N compression load. This was the case for all specimens so far and the specifications should detail this.
Patellofemoral joint testing delayed to update error catching feature of the new SimVitro software in order to accommodate virtual force transducer utilized when the femur is mounted up. There were other software issues that needed to be fixed. These came as a result of the manual process of getting specific properties from the tibiofemoral state configuration file and placing in the state file for patellofemoral testing. As a follow up, we are creating a script to automating this conversion process. This script has been unit tested, but will be validated during the next test.
- In patellofemoral joint testing, some conditions needed to be repeated as a result of load application issues and robot shut down. The last trial of these conditions should be used for future data analysis.
Tissue Mechanics
Specimen Preparation
Reference Specification: Specifications/SpecimenPreparation - Revision as of January 7, 2015
Leading Team Member: Snehal Chokhandre
Supporting Team Members: Craig Bennetts, Jack Andrish, Ahmet Erdemir
Timeline:
- Soft tissue dissection: ~2:30 PM - ~5:00 PM January 7, 2015
- Cartilage dissection: ~ AM - PM January 8, 2015
Data Location:
Protocol Deviations:
- Soft tissue dissection started late. The team decided to postpone the cartilage dissection to the following day. As a result, bone and cartilage were kept in the fridge another night for dissection in the following morning. All ligament, tendon, menisci segments and secondary tissues were dissected and frozen.
Notes:
- Jack Andrish provided a detailed dissections of the soft tissue structures of the knee, including various tendons and capsular components. Many of the secondary tissue types were identified, dissected, and stored in addition to semitendinosus, medial patellofemoral ligament, fat bursa, transverse ligament, etc.
- Per Jack Andrish, overall assessment of the specimen was healthy relative to the age of the donor.
Testing
Reference Specification:
Leading Team Member:
Supporting Team Members:
Timeline:
Data Location:
Protocol Deviations:
- None.
Notes:
- None.
Data Analysis
Registration
Spherical Markers
- Femur
<Posterior>oks006_MRG_FMR-P_AGS_01.stl
<Lateral>oks006_MRG_FMR-L_AGS_01.stl
<Medial>oks006_MRG_FMR-M_AGS_01.stl
- Tibia
<Posterior>oks006_MRG_TBR-P_AGS_01.stl
<Lateral>oks006_MRG_TBR-L_AGS_01.stl
<Medial>oks006_MRG_TBR-M_AGS_01.stl
- Patella
<Superior>oks006_MRG_PTR-S_AGS_01.stl
<Lateral>oks006_MRG_PTR-L_AGS_01.stl
<Medial>oks006_MRG_PTR-M_AGS_01.stl
Digitizing Order
- Tibia:
- Sphere 1 = Lateral
- Sphere 2 = Medial
- Sphere 3 = Posterior
- Femur:
- Sphere 1 = Lateral
- Sphere 2 = Medial
- Sphere 3 = Posterior
Joint Mechanical Testing
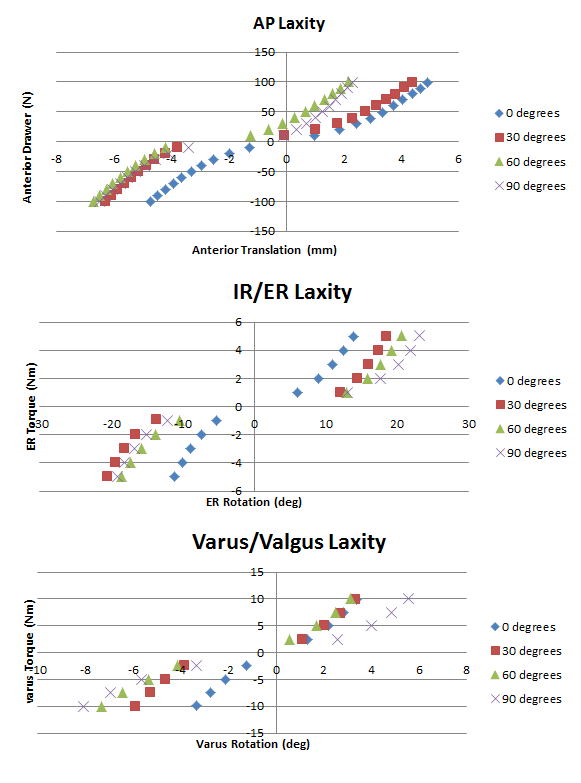
Pictured below are the Anterior Posterior laxity graph, the IR/ER laxity graph, and the Varus/Valgus laxity graph.